Cannabis extraction has been used as a blanket term for what is best described as cannabis processing. A well-designed cannabis process goes far beyond just extraction, overlapping heavily with cultivation on the front-end and product development on the back-end [1]. With this mindset, four pillars emerge as critical capabilities for developing the ideal cannabis process: Cultivation, Analytics, Extraction, and Biochemistry.
Taken alone, the purpose and value of each pillar are quite clear, but it is only when combined that each pillar can be optimized to provide their full capacities in a well-designed process. Because of this, it is best to define the goals of each pillar alone, and then determine how they can be further augmented.
Cultivation
Cultivation is the soul of any horticultural process, including cannabis production. Whether the goal is to convert flavors, pigments, or bioactive compounds into a usable form, a natural process should only harness what is provided by the raw material, in this case, the cannabis flower. That means cultivation provides a molecular feedstock for our process and, depending on our end goals, there are many requirements we may have in mind. These requirements start as simply as mass yield. Different metrics that can be used here include mass yield per square foot or per light. Taken further, this yield may be expressed based not only on mass, but the cannabinoid content of the plants grown. This could give rise to a metric like CBD or THC yield per square foot and may be more representative of a successful grow. Additionally, as scientists race to learn more about the ways individual cannabinoids and their combinations interact with the human body, cultivators will be more focused on identifying cultivars that provide unique ratios of cannabinoids and other bioactive compounds consistently. Further, research into the synergistic effect of terpenes with cannabinoids suggests that terpene content should be another goal of cultivation [2]. Lastly, and most importantly, it is critical that cultivation provides clean and safe materials downstream. This means cannabis flower free of microbial growth, heavy metals, pesticides, and other contaminants.
Analytics
Analytics provide a feedback loop at every step of cannabis production. Analytics may include Liquid Chromatography methods for cannabinoids [3], [4], [5] or Gas Chromatography methods for terpene content [3]. Analytical methods should be precise, accurate, and specific. Ideally, they can identify the compounds and their concentrations in a cannabis product. Analytics is a pillar of its own due simply to the efforts required to ensure the quality and reliability of results provided as well as ongoing optimization of methods to provide more sensitive and useful results. That said, analytics are only truly harnessed when paired with the other 3 pillars.
Extraction
Extraction is best thought of as the conversion of target molecules in cannabis raw material to a usable form. Which molecules those are is up to the goals of your product. The spectrum runs from an extract containing only a pure, isolated cannabinoid-like CBD, to an extract containing over 100 cannabinoids and terpenes in a predictable ratio. There are nearly limitless approaches to take in terms of equipment and process optimization in this space so it is critical to identify which is the best fit for the end-product [1].
Biochemistry
Biochemistry can be split into two main concepts. Plant biochemistry focuses back towards cultivation and allows a cannabis scientist to understand the complex pathways that give rise to unique ratios of bioactive molecules in the plant. Human biochemistry focuses on how those bioactive molecules interact with the human endocannabinoid system, as well as how different routes of administration may impact the pharmacokinetic delivery of those active molecules.
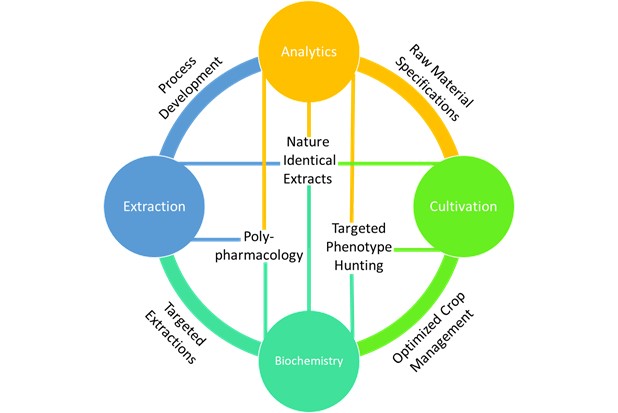
Each of the pillars require technical know-how and resources to build, but once established they can be a source of constant innovation. Fig. 1 above shows how thoroughly each of these pillars are connected. At the intersection of each pillar, unique technology platforms exist capable of driving an innovation and discovery cycle towards the development of ideal products.
For example, instead of blindly processing cannabis based on cultivar name, I could apply analytics to determine the terpene and cannabinoid content of the incoming raw materials and form specifications to ensure the ongoing quality and consistency of my products. Also, analytics can provide feedback as I tweak variables in my extraction process resulting in optimized methods. By applying plant biochemistry to my cultivation with the assistance of analytics I could begin hunting for specific phenotypes within cultivars that provide elevated levels of specific cannabinoids like CBGA or THCV. By understanding human biochemistry tied with well-characterized extracts, I can begin harnessing the overwhelmingly complex polypharmacology associated with multidrug, multitarget plant cannabinoid therapies [6]. When these pillars are harnessed effectively, we can fully characterize our unique cannabis raw material with targeted cannabinoid and terpene ratios, optimize an extraction process to ensure no loss of desirable bioactive compounds, compare our extracted product back to its source, and ensure we are delivering a safe, consistent “nature identical” extract for use in products.
With these tools in hand, we can confidently set about the task of processing safe, reliable, and well-characterized cannabis extracts for the development of world-class products.
[1] Sweeney, C. “Goal-Oriented Extraction Processes.” Cannabis Science and Technology, vol 1, 2018, pp 54-57.
[2] Russo, E. B. “Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects.” British Journal of Pharmacology, vol. 163, no. 7, 2011, pp. 1344–1364.
[3] Giese, Matthew W., et al. “Method for the Analysis of Cannabinoids and Terpenes in Cannabis.” Journal of AOAC International, vol. 98, no. 6, 2015, pp. 1503–1522.
[4] Gul W., et al. “Determination of 11 Cannabinoids in Biomass and Extracts of Different Varieties of Cannabis Using High-Performance Liquid Chromatography.” Journal of AOAC International, vol. 98, 2015, pp. 1523-1528.
[5] Mudge, E. M., et al. “Leaner and Greener Analysis of Cannabinoids.” Analytical and Bioanalytical Chemistry, vol. 409, 2017, pp. 3153-3163.
[6] Brodie, James S., et al. “Polypharmacology Shakes Hands with Complex Aetiopathology.” Trends in Pharmacological Sciences, vol. 36, no. 12, 2015, pp. 802–821.
Get daily cannabis business news updates. Subscribe
End
